How We Use Patient Data in FutureNeuro Research
Using Medical Records and other Patient Data in Health Science Research
Every visit to the doctor creates a new medical record. There’s documentation of the visit, the medications taken, medical procedures, and so much more. Medical records allow our doctors to track important information about their patients, and also allow patients to review their own medical history.
Did you know that medical records can also be used for research? With the appropriate permissions, scientists are able to assess medical records and learn important information about specific diseases or patient populations. This article will review some of the ways medical records can be used for research and how the information from medical records can be used to help understand diseases such as Epilepsy.
How are medical records used for research?
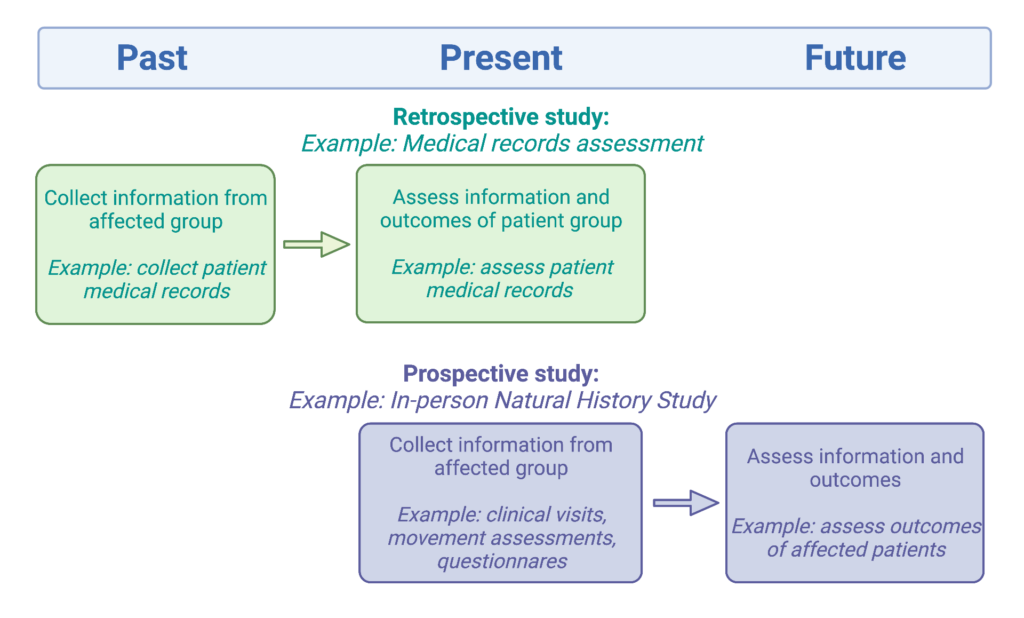
There are many different ways that medical records are used for research. We won’t be able to cover all the ways medical records are used but we will cover a few of their uses. Medical records analysis is a retrospective study, meaning the information is collected from something that has already taken place. This is in contrast to a prospective study, which follows individuals over time and watches for outcomes.
We would like to thank the TESS Research Foundation for SLC13A5 Epilepsy for kind permission to use some of their images and text in this article.
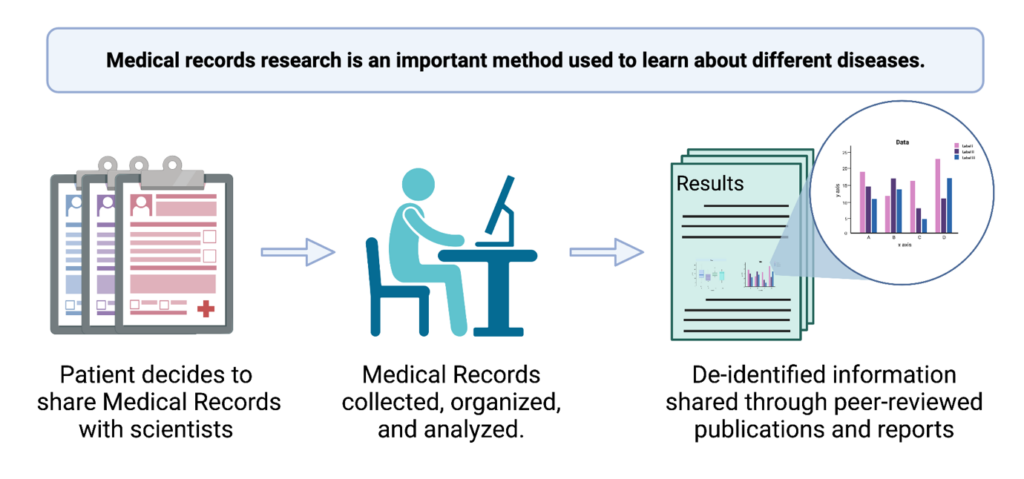
Medical records research is also easier for the patients involved in the study. There are no extra appointments or visits required, just sharing the medical records with the scientist. Once the scientist has access to the medical records, this is when they can use all the information to answer questions about a specific disease or patient population.
Sharing the medical records with the scientist is a key step for this type of study. Then, the scientist must scour all the medical records for the appropriate information. If medical records are collected from multiple patients, this can easily be 1000s of pages of information!
Sometimes there are companies that collect information and extract relevant information from the medical records to share with researchers. This requires patients to sign a consent form that allows the company to collect medical records on their behalf and also allows the company to share that information with researchers. When this information is shared, it is generally shared as de-identified data, meaning the researchers involved in the study will not be able to identify an individual patient. De-identifying data protects patient privacy.
What kinds of questions can be answered with medical records research?
Scientists can use medical records to study individual diseases by collecting medical records from patients with a specific disease such as epilepsy. Scientists can then answer questions such as:
- What does this disease look like over time?
- What are the most commonly used medications?
- How often do patients stay in the hospital?
- How often do specific symptoms occur?
- Are there biomarkers associated with the disease?
- What developmental milestones are met or missed by patients?
There is a plethora of different information captured in medical records. Asking the best questions depends on what the scientist is trying to understand. If all patients frequently undergo a specific type of procedure, this information can be particularly useful. For example, it is common to measure certain things during routine healthcare appointments. Some of these are:
- height
- weight
- blood pressure
- heart rate
These measurements would provide a significant amount of information and researchers could use it to track changes over time. Depending on the type of procedures, there may be other types of information such as blood analysis, EEGs or MRIs captured in the medical records. All of this information provides crucial insight into understanding any disease. The next step becomes extracting the information, organizing the information, and then assessing it for all patients. This can be a challenging step that can provide important information about patient populations.
Why are medical records important for research in rare disease?
Having access to medical records is extremely powerful. With more data, scientists are able to make stronger conclusions that help guide the development of treatments. This is often challenging for rare disease communities with a limited number of patients. For example, sometimes a majority of the patients are young so scientists don’t know what happens to patients as they get older. Having medical records from patients across many different ages is extremely beneficial because this allows scientists to look at how the disease changes over time.
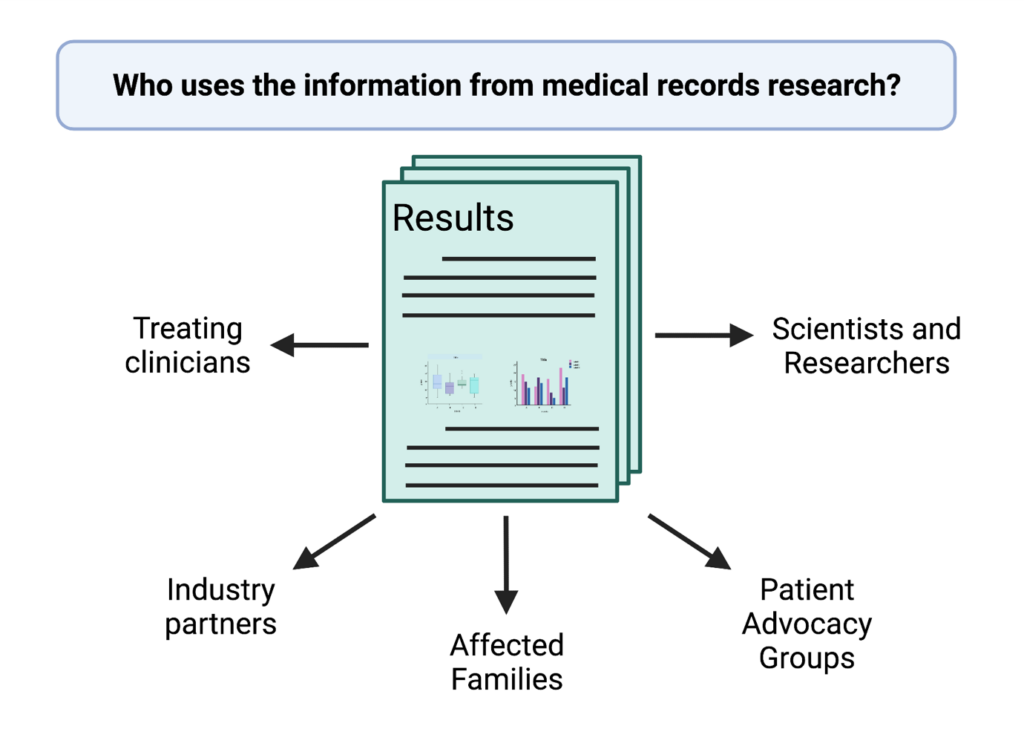
Medical records research is also important for rare diseases because it allows scientists to share important disease information with the treating clinicians, industry partners (who may be interested in developing a therapeutic to help treat a disease), scientists studying the disease, and also with affected families. This information helps provide a better understanding of the disease and improves treatments, as well as facilitates the development of new treatments. Sharing this information also helps to keep research patient-centred, because it focuses on the needs of the patient.
How are medical records being used for FutureNeuro neurological research?
FutureNeuro is very committed to combining medical records research with genomic testing results. Currently our work in this area is focussed on epilepsy and seizure disorders but over time we hope to expand this type of work to help bring advanced diagnostic ability to the clinical setting for a broader range of neurologically significant conditions.
An exciting part of our work involves receiving de-identified data from genetic tests taken from, for example, patients at Children’s Health Ireland, St James’s Hospital, Beaumont Hospital. As soon as we receive data we start analysis right away.
All of our studies have ethical approval from participating hospitals and a number of these studies also have approval from the Health Research Consent Declaration Committee (HRCDC). It is recognised – as it is in other countries – that sometimes, in limited situations, obtaining consent from patients themselves will not always be possible and that the public interest of doing the research significantly outweighs the need for explicit consent. It is in cases like this that the HRCDC has a decision making role. We take very seriously our privileged position of trust to be granted approval from the HRCDC and hospital ethics committees to engage in public interest research.
Currently we are working on two large dataset-based studies that have this type of special permission:
EPIDIVE: Longitudinal analysis of clinical markers of response to treatment in people with epilepsy. The aim of this project is to discover factors associated with a positive response to seizure control sustained over time by looking at large volumes of patient records on anti-epileptic drug treatment, vagus nerve stimulation and epilepsy surgery.
HPO: A description of the evolution of phenotype in epilepsy from paediatrics through adulthood and old age. The aim of this project is to track characteristics of patients’ epilepsy to see if there are any significant variations in the frequency of occurrence of those characteristics over the course of their illness.
By comparing Irish data with international data we can increase the power of the analysis that can be performed but there is much work to be done to ensure that there is a common and consistent understanding of the clinical terms used in the analyses. To put it simply, we need to be comparing like with like so ensuring that the Irish data is analysis-ready requires a lot of work by our researchers and clinicians.
For these studies we use patient data from electronic epilepsy patient records from Beaumont and St. James’s Hospitals. The data used is de-identified, we ensure that researchers do not have access to the personal information of individual patients.
As our work progresses we wish to share this information with the patients and the public as well as the clinical and scientific community. We will publish our results in a peer-reviewed publications and present them at research conferences.
We are also committed to sharing our results in non-technical, accessible formats that will be meaningful to the wider community. We will make this information available on our website, social media channels and share it directly via our Education and Engagement mailing list.
If you would like to receive emails from us or get involved in our education and engagement activities please register here.
Your Data Rights – find out more here.

Current Clinical Studies
FutureNeuro has a large number of ongoing pre-clinical and clinical research studies.
Digital Health
Our goal is to lead the way in developing personalised, accessible, proactive, and preventive healthcare solutions, enabled by digital technologies, and specifically tailored for those impacted by neurological and neuropsychiatric diseases.